-
Methane (CH4) is one of the main greenhouse gases with a strong absorption band in the infrared region, and its contribution to global warming accounts for about 20% (IPCC, 2013; Dutta et al., 2015). The global warming potential of CH4 gas in 100 years is 25–32 times that of carbon dioxide (CO2) (IPCC, 2007; Solomon, 2007; Neubauer and Megonigal, 2015). As of 2019, the global atmospheric concentration of CH4 is 1.877 ± 0.002 μL L−1, which is 2.6 times that of the preindustrial period, with an average growth rate of 0.43% in the last decade (WMO, 2020). The increase in anthropogenic CH4 sources (Solomon, 2007; Barrie et al., 2009) and the decrease in natural CH4 sinks (FAO, 2010) are the main reasons for the rapid increase in the CH4 concentration in the atmosphere. If CH4 emissions can be stabilized, the global temperature rise can be reduced by 25% (Thompson et al., 1992). Therefore, research on global CH4 sources/sinks has attracted the attention of environmental scientists (Carmichael et al., 2014).
Vegetation is an important factor in global climate change and plays a key role in the complex interactions between the land surface and the atmosphere (Hong and Lakshmi, 2005). Studies have shown that different vegetation has different effects on soil CH4 gas (Dalal and Allen, 2008; Kim et al., 2016; Oertel et al., 2016; Plain et al., 2019; Zhang et al., 2019; Chai et al., 2020; Ma et al., 2020), with production, oxidation, and transport being the three most important processes that control soil CH4 fluxes (Bubier et al., 1993; Cao et al., 1996; Harazono et al., 2006; Lai, 2009; Von Fischer et al., 2010; Brummell et al., 2012). The composition and density of plant communities are extremely important in controlling soil CH4 fluxes (Sebacher et al., 1985; Schimel, 1995; Shannon et al., 1996; Ding et al., 2004; Mills et al., 2013; Lai et al., 2014a, b). Vegetation affects soil CH4 fluxes by changing the physicochemical properties of the soil (Bhandral et al., 2007; De La Bárcena et al., 2014), and a well-developed root system will loosen the compaction of the soil, thereby creating an aerobic environment that affects the flow of water and gases and influences the uptake and diffusion of soil CH4 (Bhandral et al., 2007; Shi et al., 2013). Vegetation can have a key impact on soil CH4 flux by providing organic substrate and increasing the transport of CH4 (Jones et al., 1987; Schimel, 1995; Ström et al., 2003; Harazono et al., 2006; Von Fischer and Hedin, 2007; Bhullar et al., 2013a). After CH4 production, the aeration tissues of herbaceous plants facilitate CH4 transport by providing important pathways for CH4 movement between soil and atmosphere (Joabsson et al., 1999; Ström et al., 2003; Whalen, 2005; Brummell et al., 2012; Bhullar et al., 2013a, b), allowing CH4 to bypass the oxidation layer in the soil where it would otherwise be reoxidized (Whalen and Reeburgh, 1990; Jespersen et al., 1998; Frenzel and Karofeld, 2000; Heilman and Carlton, 2001; Inubushi et al., 2001; Joabsson and Christensen, 2001; Ström et al., 2005; Wilson and Humphreys, 2010). Also, the abundance of vegetation affects the growth and activity of aerobic microorganisms, and thus the ability of CH4 to diffuse from soil to atmosphere (Schimel, 1995; Rusch and Rennenberg, 1998). Moreover, plants can also help the competitive process of CH4 oxidation by transporting oxygen to their roots, which can support CH4 reproduction when it is released into the surrounding soil (Sebacher et al., 1985; Conrad, 1996; Harazono et al., 2006). Therefore, it is crucial to understand the mechanisms by which vegetation affects soil CH4 fluxes.
The processes controlling soil CH4 fluxes are closely coupled to surrounding environmental conditions (Jones et al., 1987; Harriss and Frolking, 1992; Harazono et al., 2006; Von Fischer et al., 2010) and are community-wide (Billings, 1952). Therefore, the temporal variability of soil CH4 fluxes under different vegetation is controlled by different factors. For example, in the Prince Albert National Park, Saskatchewan, soil CH4 emissions are the highest in boreal aspen forest from late July to mid-September, and there are strong correlations between soil CH4 fluxes and soil temperature (Simpson et al., 1997). The seasonal peak of soil CH4 uptake in artificial grassland soils in the semi-arid region of Inner Mongolia occurs in the season when rainfall is more concentrated, and the factors affecting the seasonal variation of soil CH4 uptake are mainly soil moisture and soil temperature (Wang, 2014). In a typical Mediterranean climate zone, soil CH4 uptake fluxes reach a maximum in summer and a minimum in winter, and have a daily variation of small uptake fluxes during the day and large uptake fluxes at night (Fernández-Duque et al., 2020). Soil CH4 is absorbed during the growing season and emitted during the non-growing season in the Huoditang Forest Region of the Qinling Mountains, with large seasonal fluctuations. Overly low temperatures inhibiting CH4 oxidation is the main reason for the appearance of soil CH4 emissions during the non-growing season (Liu et al., 2019). In the Abbey Lake area of the arid zone, the soil CH4 emission from the abandoned land has a more obvious diurnal variation with a single or double-peaked curve, and moisture and temperature are the most important environmental factors affecting the variation of soil CH4 emission from the abandoned land during the growing season (Sun et al., 2012). Therefore, it has become increasingly important to explore the main factors that play a decisive role in soil CH4 fluxes.
Loess Hilly Region (LHR) is the most fragile and degraded area in China (Tang et al., 1993) and a key area of ecological restoration. In recent years, in order to control soil erosion effectively and realize ecological restoration and its virtuous cycle, a project known as “the Conversion of Farmland to Forests and Grasses Program” has been implemented in this area (Wu et al., 2018). This also allows the local vegetation to enter a relatively undisturbed recovery process. However, most studies on the vegetation in LHR have focused on soil physical properties (Wu et al., 2018), soil nutrients (An et al., 2006; Wang et al., 2020), enzyme activities (Yang et al., 2015; Wang et al., 2020), the soil organic carbon (SOC) pool, etc., with relatively few reports on the soil CH4 flux under the different vegetation in this region. Therefore, it is very important, for regional and national carbon sink assessment in China, to increase understanding on the response of soil CH4 flux to vegetation differences.
In this study, we investigated the effects of different plant community types (Medicago sativa grassland, Xanthoceras sorbifolium forestland, Caragana korshinskii bushland, Hippophae rhamnoides shrubland, and Stipa bungeana grassland) on soil CH4 flux in LHR from November 2017 to October 2019 using a closed static chamber method. The main aims of the study were to: (1) determine the differences in soil CH4 fluxes among different plant community types in LHR; and (2) identify the main environmental factors affecting soil CH4 fluxes. We hypothesized that, among the five plant community types in LHR, (1) there would be significant differences in soil CH4 fluxes among plant community types; (2) the Stipa bungeana grassland, which is dominated by herbaceous plants with a clear numerical predominance, would be the strongest soil CH4 sink among the five plant community types; and (3) soil temperature and soil moisture would influence the temporal variability of soil CH4 fluxes.
-
The study area was Anjiagou Watershed (35°33′02″–35°35′29″N, 104°38′13″–104°40′25″E), located in Anjiapo Village, Fengxiang Town, Dingxi City, Gansu Province, which belongs to the typical semi-arid loess hilly gully area of the fifth subregion (Fig. 1). The watershed covers an area of 8.54 ha (1 ha = 0.01 km2), with elevation between 1900 m and 2250 m. The area has a temperate continental monsoon climate with an average annual temperature of 6.3°C, average annual precipitation of 427 mm, and evaporation level of 1500 mm, with concentrated and seasonal precipitation (more than 60%) occurring from May to September, mainly in the form of heavy rainfall (Fig. 2). The main soil types are loess and river saline soils (Gong et al., 2007). Before the 1990s, the area was deforested and cleared, and in 1999, the Conversion of Farmland to Forests and Grasses Program was officially launched to plant vegetation in the ratio of 1:2:7 with trees, shrubs, and herbs (Wang and Bennett, 2008). In the early stage of afforestation, the survival rate of the fallow forest and grass was improved by artificial irrigation and replanting measures. The artificially restored vegetation species in the watershed were X. sorbifolium, C. korshinskii, and H. rhamnoides. The herbaceous species were mainly M. sativa (Table 1). Xanthoceras sorbifolium (family: Sapindaceae) is a small- to medium-sized tree endemic to northern China and is an emerging oil crop used for advanced biofuels, functional foods, and pharmaceutical and cosmetic applications (Zhou and Cai, 2021). Caragana korshinskii (family: Leguminosae) is a perennial leguminous shrub widely distributed in arid and semi-arid regions of Eurasia, with important environmental benefits and economic value for sand fixation and water retention (Bai et al., 2017). Hippophae rhamnoides (family: Elaeagnaceae) is a tree species with drought, sand, and salt tolerance, which is planted in large quantities in northwestern China for desertification control and has a high ecological value (Wei et al., 2019). Medicago sativa (family: Leguminosae) is a perennial herbaceous plant with strong drought resistance and adaptation to poor soils, which is widely planted in the region (Ji et al., 2020).

Figure 1. Map of the study area: XS, Xanthoceras sorbifolium forestland; SB, Stipa bungeana grassland; CK, Caragana korshinskii bushland; HR, Hippophae rhamnoides shrubland; MS, Medicago sativa grassland.

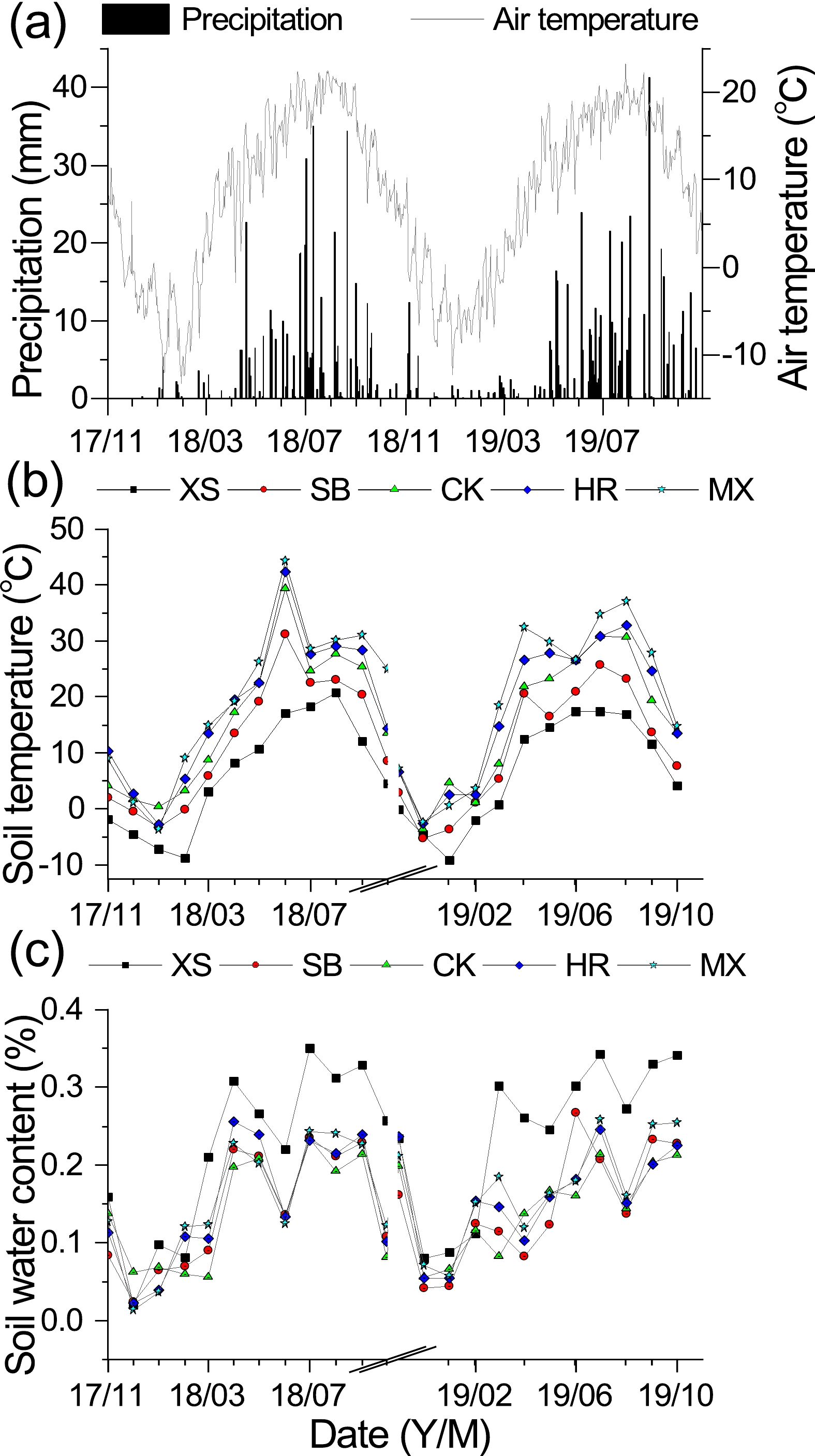
Figure 2. Temporal patterns of (a) air temperature and precipitation of the study area, and (b) soil temperature and (c) soil water content in the top 10 cm of the five plant community types: XS, Xanthoceras sorbifolium forestland; SB, Stipa bungeana grassland; CK, Caragana korshinskii bushland; HR, Hippophae rhamnoides shrubland; MS, Medicago sativa grassland.
Plot Area (m2) Longitude and latitude Elevation (m) Main species Coverage (%) Height (cm) MS 20 × 20 104°39′1.82′′E
35°34′48.07′′N1990 Medicago sativa (90%), Stipa bungeana, Artemisia
lavandulifolia90.06±1.58b 62.23±0.23d XS 20 × 20 104°39′10.62′′E
35°34′45.08′′N2018 Xanthoceras sorbifolium (86%), Bupleurum chinense,
Gentiana macrophylla, Leontopodium leontopodioides95.32±2.35c 76.45±2.54e CK 20 × 20 104°39′1.51′′E
35°34′45.00′′N1999 Caragana korshinskii (42%), Potentilla chinensis,
Picris hieracioides50.43±5.45a 16.00±0.79a HR 20 × 20 104°39′0.18′′E
35°34′47.32′′N1998 Hippophae rhamnoides (81%), Leontopodium
leontopodioides, Artemisia annua89.26±0.78b 54.89±1.23c SB 20 × 20 104°39′3.05′′E
35°34′45.48′′N2008 Stipa bungeana (92%), Plantago asiatica, Setaria
viridis, Leymus secalinus98.23±0.63c 35.93±0.44b Notes: XS, Xanthoceras sorbifolium forestland; SB, Stipa bungeana grassland; CK, Caragana korshinskii bushland; HR, Hippophae rhamnoides shrubland; MS, Medicago sativa grassland. The percentage of the main species is the coverage of the dominant species. Values are presented as mean ± standard error. Superscript letters indicate significant differences between plant community types (P < 0.05). Table 1. Basic information on the different plant community types at the experimental sites.
-
In September 2017, the ecosystems in the study area were classified into several types according to the different vegetation restoration types and the dominant species of the plant community, and five representative plant community types were selected as observations for the measurement of differences in soil CH4 dynamics: X. sorbifolium forestland (XS), Stipa bungeana grassland (SB), C. korshinskii bushland (CK), H. rhamnoides shrubland (HR), and M. sativa grassland (MS). XS, CK, and HR were restored to woodland, mallow shrub community, and buckthorn shrub community, respectively, in 1999 through the implementation of the Conversion of Farmland to Forests and Grasses Program and manual management, without any further management measures. MS was abandoned in 2015 on the basis of cultivated land, planted with M. sativa and enclosed, and no further management measures were taken after survival. SB was abandoned in 1999 and naturally restored to an herbaceous community without any further management measures. Regarding the slope orientation, XS was located on the northeast slope, CK on the northwest slope, HR and MX on the north slope, and SB in the no-slope orientation zone. All sampling plots were set on sloping (but not significantly) arable land (except HR, which had a slope of about 5°). According to data from the local Hydrological and Water Resources Survey, the depth of the water table in the area during the experiment was about 100 m. Full details of the experimental sites are presented in Table 1 and Fig. 3. A completely randomized design was used, and three randomly selected sampling plots (7 m × 7 m) were taken from each plant community type, with a buffer zone of at least 5 m between each sampling plot, for a total of 15 sampling plots.
-
Stainless steel bases (50 cm × 50 cm × 15 cm, L × W × H) were laid out in September 2017 within the 15 selected sampling plots following the method in Ma et al. (2020) (Fig. 4). Soil CH4 fluxes were sampled twice a month (biweekly) during the growing season (April to October) and once a month (mid-month) during the non-growing season (November to March) from November 2017 to October 2019 (i.e., 24 months) (Xia et al., 2015). All samples were collected between 0900 local standard time (LST, LST=UTC+8 hours) and 1200 LST. The fluxes measured during this time are considered to be representative of the daily average fluxes (Wang and Wang, 2003; Lin et al., 2009; Ma et al., 2018). After the chamber was closed, air samples (five in total) were taken from inside the chamber at 10-min intervals (at minutes 0, 10, 20, 30, and 40) using a 100 mL polypropylene syringe equipped with a three-way plug valve (Ma et al., 2020). The air samples were transferred to pre-evacuated E-Switch aluminum foil composite film gas sampling bags (Shanghai Shenyuan Scientific Instruments Co., Ltd., Shanghai, China) via the three-way plug valve. Within 48 h, the samples were returned to the laboratory for analysis of CH4 concentrations using a gas chromatograph (YiMeng A90, Changzhou Ban’nuo Instruments Co., Ltd., China). While collecting air samples, the temperature in the chamber and the soil temperature (T10) and soil water content (SWC10) at 10 cm above ground were measured using a portable digital thermometer (JM624, Jinming Instrument Co., Tianjing, China) and a temperature and humidity sensor (GS3, METER Group, Pullman, WA, USA), respectively. Air temperature and precipitation data were obtained from a local weather station located in Anjiagou Watershed. The weather station was set up near the SB sampling plot (Fig. 1), and all sampling plots were within a 500 m radius. Thus, air temperature and precipitation were not subjected to any correction.

Figure 4. Photographs of the chamber: (a) top view and (b) side view of the sampling chamber; (c) side view and (d) top view of the stainless steel base.
The CH4 gas flux was calculated as
where F is the gas flow rate (units: mg m−2 h−1),
${\rm d}C/{\rm d}t$ is the gradient of the time series of CH4 concentration (units: ppmv h−1) at the time of sampling, M is the molar mass (units: g mol−1) of the measured gas, P is the pressure (units: hPa) at the sampling plot, and T is the temperature in the chamber (units: K) at the time of sampling. V0, P0, and T0 are the molar volumes (L mol−1) of the gas at standard conditions, air pressure (units: hPa), and absolute temperature (units: K), respectively, and H (units: m) is the height of the sampling box above the ground (Song et al., 2009). The data can be subjected to later calculations only if the goodness-of-fit (R2) of the linear regression results for four or five samples is ≥ 0.80 (Ma et al., 2020). Mean fluxes are expressed as the mean and standard deviation of three replicates.The cumulative flux of CH4 gas was estimated as follows:
where i and i + 1 are the ith and ith + 1 observations, respectively; Fi is the CH4 flux (units: mg m−2 h−1) of the ith observation; and Di is the Julian date of the ith observation.
-
In September 2017, the relative coverage, plant height, and dominant species were measured randomly in 15 sampling plots (50 cm × 50 cm) (Ma et al., 2018). At the same time, soil samples were taken from the 0–10 cm layer using a soil auger (diameter: 5 cm) following the diagonal five-point method (four points selected at each end of the “X” and one point at the intersection) (Wang et al., 2020). Five soil samples from the same soil layer in each plot were mixed to form one soil sample, for a total of 15 soil samples. After removing debris such as stones and residual roots, the samples were dried and put into self-sealing bags through 100-mesh soil sieves for determination of the soil pH value, SOC, and total nitrogen (TN).
The soil total porosity (STP) of the 0–10 cm layer was measured using the ring knife method (volume: 100 cm3) (Wang et al., 2020). Soil pH was measured with a pH meter (PHS-3S, INESA Scientific Instrument Co., Shanghai, China), and the soil water ratio of suspension was 1:5 (Wu and Mu, 2019). SOC and TN were determined by referring to the method of Wu et al. (2020).
-
The data were organized using Microsoft Excel 2019, and all data were statistically analyzed using SPSS 22.0. One-way analysis of variance (Duncan’s Multiple Range Test, P < 0.05) was used to determine the differences in vegetation properties (coverage, height), soil CH4 fluxes, and soil characteristics [pH, STP, SOC, TN, ratio of carbon to nitrogen (C/N), T10, SWC10] among plant community types. General linear model (GLM) analysis and analysis of covariance (ANCOVA) were used to compare the interaction and effects of different plant community types (PCT) and soil temperature/soil water content on soil CH4. The relationships between environmental variables (T10, SWC10) and soil CH4 fluxes in the five plant community types were analyzed using Pearson correlation analysis (two-tailed). The interrelationships between T10, SWC10, and soil CH4 fluxes in the five plant community types were analyzed using a linear regression method.
-
The vegetation properties (dominant species, vegetation cover, plant height) varied clearly at the five sites (Table 1). The plant community type significantly affected the physical properties of the soil (STP, T10, SWC10) throughout the experimental period (P < 0.05, Table 2). The STP of MS was significantly lower than that of the other four plant community types; the mean T10 of the five plant community types from 0 cm to 10 cm showed a trend of XS < SB < CK < HR < MS; and the variation in 10 cm soil SWC ranged from 14.0% to 24.3% (Table 2), showing a trend of CK < SB < HR < MS < XS (Table 2). The average T10 and SWC10 from 0 cm to 10 cm for all five plant community types showed a trend that the growing season was greater than the non-growing season (Figs. 2b and c). The SOC of MS was significantly higher than that of the other four plant community types (P < 0.05). The soil TN of XS was significantly higher than that of the other plant community types, and the soil C/N of XS, CK, and HR was significantly lower than that of the other two plant community types.
Plot STP (%) T10 (°C) SWC10 (%) pH SOC (g kg−1) TN (g kg−1) C/N MS 49.45 ± 0.42a 19.68 ± 2.08c 0.17 ± 0.01a 6.75 ± 0.16a 11.08 ± 0.32d 0.45 ± 0.05b 25.73 ± 3.84b XS 73.39 ± 3.16c 6.75 ± 1.46a 0.24 ± 0.01b 6.92 ± 0.22a 10.58 ± 0.09cd 0.77 ± 0.00c 13.68 ± 0.11a CK 71.06 ± 2.24c 15.80 ± 1.80bc 0.14 ± 0.01a 6.58 ± 0.34a 5.45 ± 0.55a 0.29 ± 0.01a 18.86 ± 1.88a HR 62.85 ± 0.34b 17.81 ± 1.88bc 0.16 ± 0.01a 6.67 ± 0.15a 7.91 ± 0.21b 0.45 ± 0.00b 17.71 ± 0.54a SB 68.36 ± 1.03bc 11.57 ± 1.62ab 0.15 ± 0.01a 7.00 ± 0.41a 9.57 ± 0.30c 0.32 ± 0.00a 29.92 ± 0.86b Notes: STP, soil total porosity; SOC, soil organic carbon; TN, total nitrogen; C/N, carbon nitrogen ratio; T10, soil temperature of top 10 cm; SWC10, soil moisture of top 10 cm. Superscript letters indicate significant differences between plant community types (P < 0.05). Table 2. Physicochemical properties of soil (0–10 cm depth) from sites with different plant community types.
-
Throughout the observation period, soil CH4 fluxes of the five different plant community types showed net uptake, with fluxes ranging from −0.061 mgC m−2 h−1 to −0.086 mgC m−2 h−1. The soil CH4 fluxes differed significantly (P < 0.05) among the different plant community types (Fig. 5), and the average soil CH4 fluxes of SB were 1.29, 1.42, 1.20, and 1.08 times higher than those of MS, XS, CK, and HR, respectively. The cumulative soil CH4 fluxes of the five different plant community types ranged from 14.986 kgC ha−1 to 19.796 kgC ha−1 throughout the observation period, showing a trend of XS < MS < CK < HR < SB (Table 3).
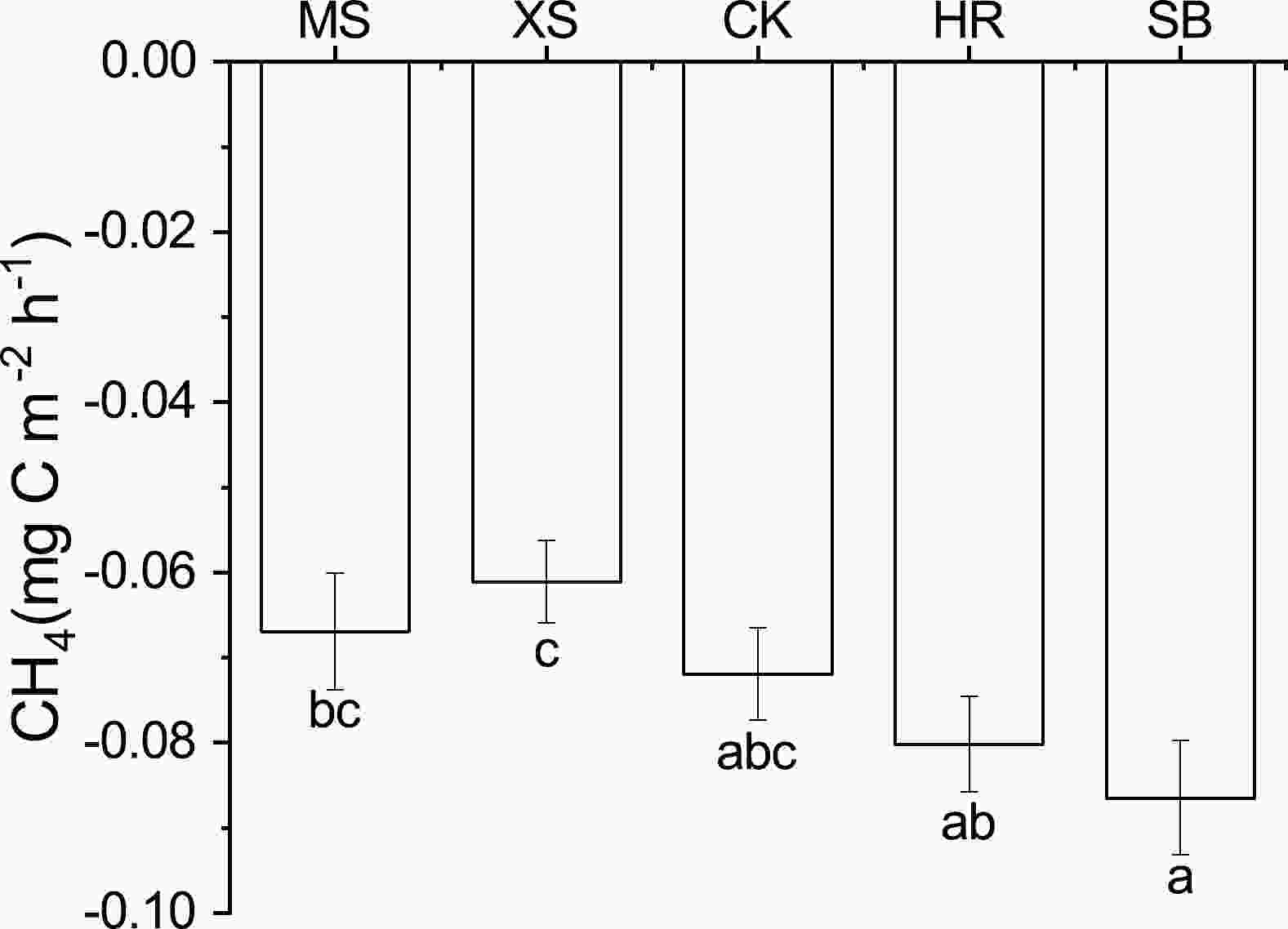
Figure 5. Annual average soil CH4 flux under different plant community types: MS, Medicago sativa grassland; XS, Xanthoceras sorbifolium forestland; CK, Caragana korshinskii bushland; HR, Hippophae rhamnoides shrubland; SB, Stipa bungeana grassland. Lowercase letters indicate statistically significant differences within observations among different plant community types (P = 0.05).
MS (kgC ha−1) XS (kgC ha−1) CK (kgC ha−1) HR (kgC ha−1) SB (kgC ha−1) Y1 −5.943 ± 0.743 −7.532 ± 0.500 −7.803 ± 0.499 −8.938 ± 0.956 −8.554 ± 1.450 Y2 −9.330 ± 0.475 −7.454 ± 0.524 −8.781 ± 0.728 −9.724 ± 0.426 −11.241 ± 0.449 Total −15.273 ± 0.295 −14.986 ± 0.500 −16.585 ± 1.127 −18.662 ± 0.737 −19.796 ± 1.566 Notes: Values are presented as mean ± standard error. Y1, November 2017 to October 2018; Y2, November 2018 to October 2019. Table 3. Cumulative estimates of soil CH4 uptake under different plant community types.
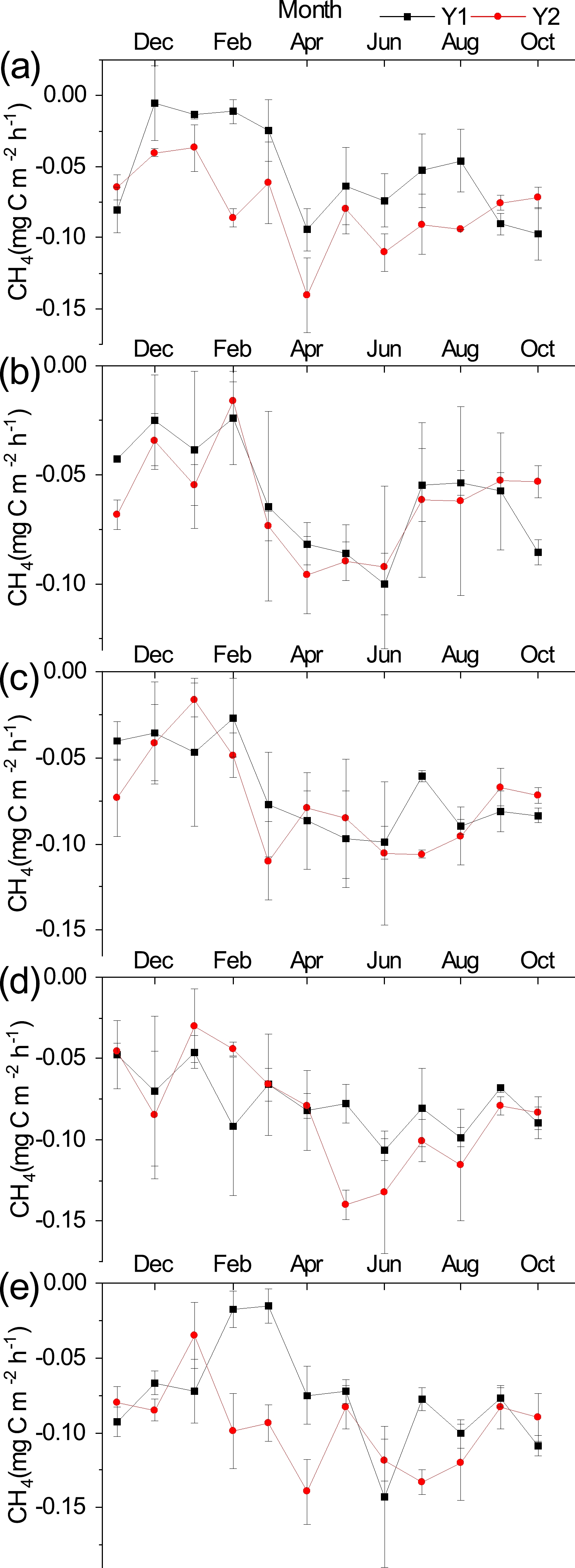
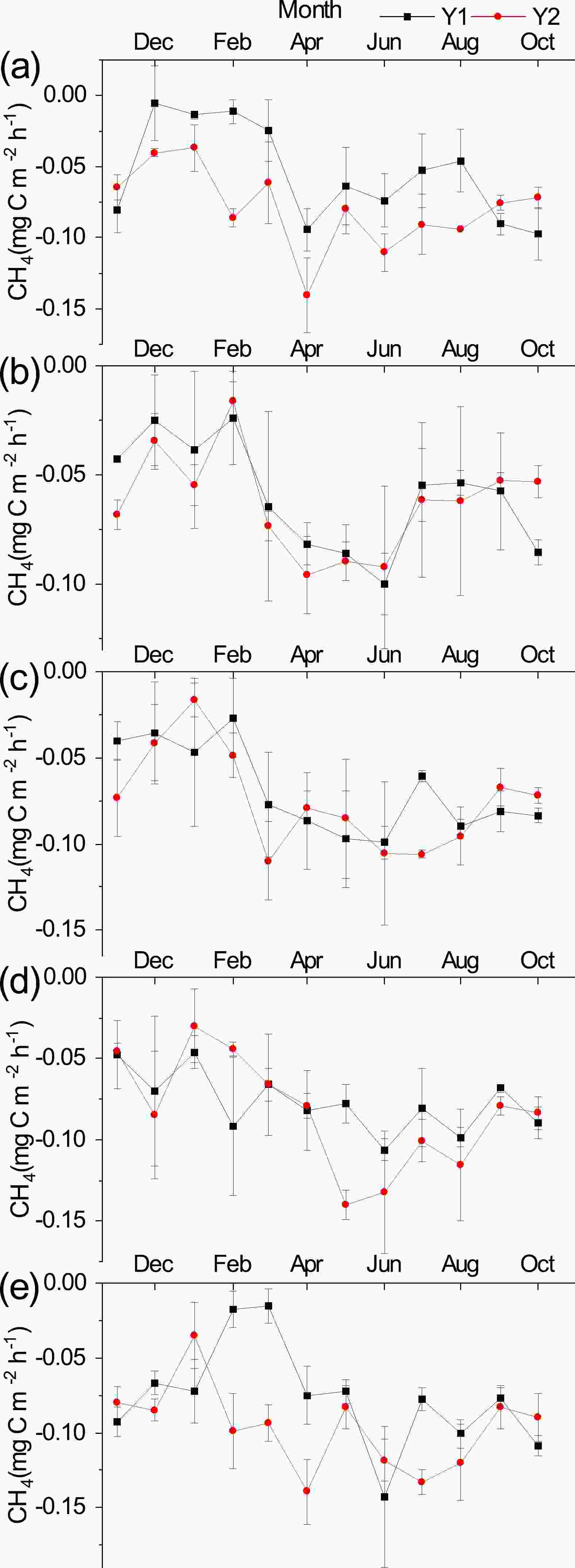
Throughout the observation period, there were significant seasonal variations in soil CH4 fluxes among the five different plant community types (Fig. 6), with soil CH4 uptake occurring mainly during the growing season [SB (−0.101 ± 0.007 mgC m−2 h−1) > HR > CK > MS > XS (−0.073 ± 0.005 mgC m−2 h−1)]. In addition, soil CH4 uptake began to decrease with fluctuation in November and reached its lowest value in February, after which the soil CH4 flux gradually increased in volatility and reached a peak in April–June.
-
GLM analysis of plant community type and soil CH4 flux showed that the interaction terms for PCT × T10 and PCT × SWC10 were not significantly different for soil CH4 fluxes (P > 0.05, Table 4), indicating that there were no interactive effects of plant community type and T10/SWC10 on soil CH4 flux. The ANCOVA of plant community type and soil CH4 flux showed that excluding the effect of T10, there was a significant effect of plant community type on soil CH4 flux (P < 0.05); and excluding the effect of SWC10, there was a significant effect of plant community type on soil CH4 flux (P < 0.05) (Table 4).
GLM analysis ANCOVA analysis Fixed factors F Sig. Fixed factors F Sig. PCT 1.353 0.251 PCT 60.392 0.000 T10 55.539 0.000 T10 4.841 0.001 PCT × T10 0.803 0.524 − − − PCT 1.579 0.181 PCT 4.121 0.044 SWC10 3.844 0.051 SWC10 3.153 0.015 PCT × SWC10 0.324 0.862 − − − Notes: Soil CH4 fluxes were used as dependent variables, while the corresponding SWC10 or T10 were covariates. The fixed factor was plant community type (PCT). The F-value is the ratio of the two mean squares (effect term/error term). Significant values (P < 0.05) are indicated in bold. Table 4. Results of GLM analysis and ANCOVA.
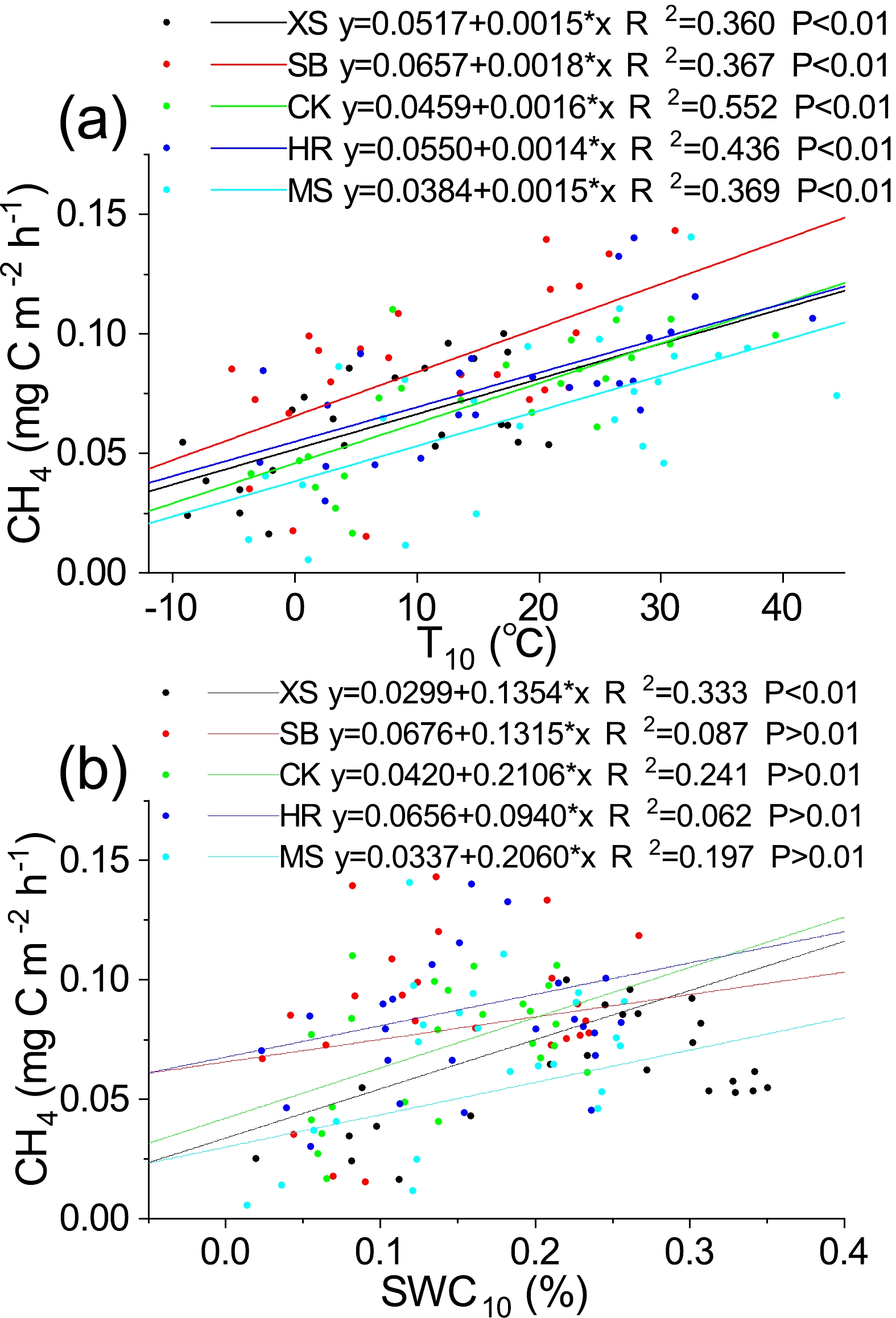
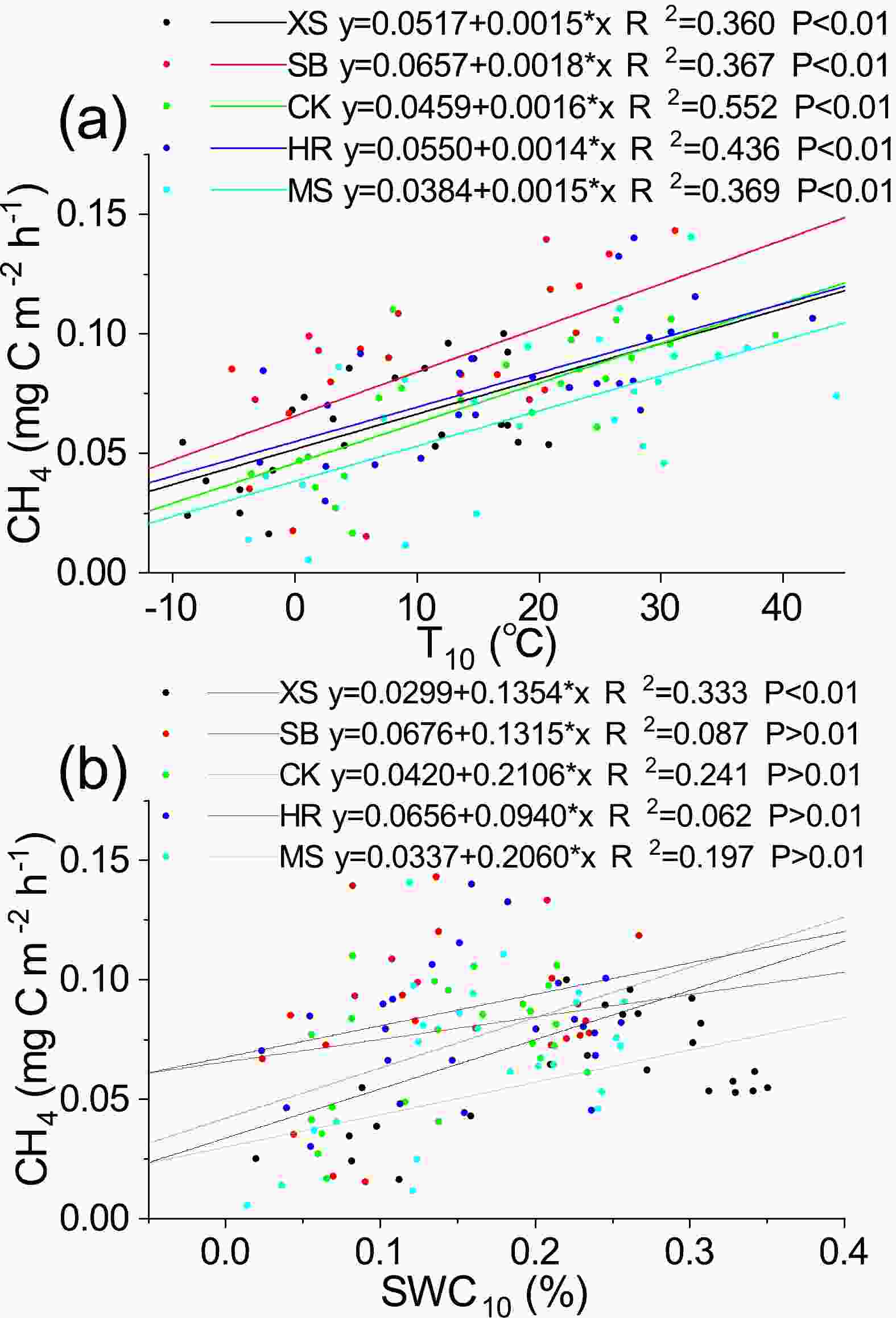
There was a highly significant positive correlation (R2 = 0.60–0.74, P < 0.01) between soil CH4 flux and T10 under different plant community types (Table 5). There was also a significant positive correlation (R2 = 0.44–0.57, P < 0.05) between soil CH4 flux and SWC10 in MS, XS, and CK (Table 5). The results of linear regression analysis for the different environmental variables under different vegetation community types showed that the variations in T10 and SWC10 explained 36.0%–55.2% and 6.18%–33.3% of the temporal variation in soil CH4, respectively (P < 0.01) (Fig. 7).
Plot T10 SWC10 MS Pearson 0.607** 0.444* Sig. (two-tailed) 0.002 0.030 XS Pearson 0.600** 0.577** Sig. (two-tailed) 0.002 0.003 CK Pearson 0.743** 0.491* Sig. (two-tailed) 0.000 0.015 HR Pearson 0.660** 0.249 Sig. (two-tailed) 0.000 0.241 SB Pearson 0.606** 0.295 Sig. (two-tailed) 0.002 0.162 Notes: A double asterisk (**) indicates significance at p < 0.01, while a single asterisk (*) indicates significance at p < 0.05. T10, soil temperature of top 10 cm; SWC10, soil water content of top 10 cm. Negative fluxes values indicating absorption (sinks) have been converted to positive values to facilitate the analysis. Table 5. Pearson correlation coefficients and their two-tailed significance between environmental factors (T10 and SWC10) and soil CH4 flux under different plant community types during November 2017 to October 2019.
-
In this study, all five plant community types were sinks of soil CH4 in LHR, with fluxes between −0.061 mgC m−2 h−1 and −0.086 mgC m−2 h−1 (Fig. 5), which is consistent with the finding of Zhao et al. (2018). The study by Wu and Mu (2019) in mountainous regions in Northeast China showed that soil CH4 fluxes in planted forests ranged between −0.048 mgC m−2 h−1 and −0.077 mgC m−2 h−1, which is significantly lower than the results of the present study and related to its lower mean annual temperature. Pihlatie et al. (2017), in boreal alpine forest, reported soil CH4 fluxes of between −0.054 mgC m−2 h−1 and −0.096 mgC m−2 h−1, which are higher than the results of this study owing to the fact that the mosses play an important role in the forest floor CH4 exchange. There were significant differences in soil CH4 flux among the five different plant community types (P < 0.05, Fig. 5), with the maximum value occurring in SB—a result similar to that of Pihlatie et al. (2017) and Zhao et al. (2018). Methane production tends to occur deep in the soil in anaerobic environments (Yang et al., 2014, 2019). In this study, the groundwater table was low, the soil envelope was thick (Chen, 2009), and most of the soils with different plant community types were well ventilated (Table 2), providing favorable oxygen conditions for the oxidation process of soil CH4 (Lombardi et al., 1997). Therefore, soil CH4 uptake dominated in this study area. In the present study, the vegetation cover of the grassland was much higher than that of the woodland, and the dense root system of the soil surface was conducive to the creation of an aerobic environment, thus increasing the uptake of soil CH4 (Shi et al., 2013). Also, larger porosity affects the rate of CH4 oxidation and methanotrophs abundance, which in turn affects the transport of CH4 in the soil (De La Bárcena et al., 2014). Added to this, the diffusion of herbaceous aeration tissues is the main pathway for gas transport, and more oxygen diffuses into the soil through the aeration tissues, which can affect the growth and activity of aerobic microorganisms and increase the oxidation of methane (Schimel, 1995; Rusch and Rennenberg, 1998). This is consistent with the results of the present study, where the soil CH4 flux of SB was significantly higher than that of other plant community types (P < 0.05, Fig. 5). This result is similar to previous findings (Schimel, 1995; Rusch and Rennenberg, 1998; Bhandral et al., 2007; Zhao et al., 2018). In SB, the number of herbaceous plants was significantly higher than that in other plant community types (Table 1), and their subsurface aeration tissues were also higher, resulting in significantly higher soil CH4 uptake in SB than in other plant community types (P < 0.05, Fig. 3a). In contrast, the soil CH4 uptake of XS was significantly lower than that of HR and SB (P < 0.05, Fig. 5) because of the higher leaf area and related shading of the soil by tall vegetation (Kim, 2013; Oertel et al., 2016), which made the T10 in XS significantly lower than that of HR and SB (Table 2). The temperatures were significantly different under different vegetation coverages (P < 0.05, Table 2), which would have limited the growth and reproduction and activity of soil microorganisms and soil enzymes (Luan et al., 2016), in turn affecting soil CH4 uptake. The decrease in temperature is accompanied by a decrease in evaporation and an increase in soil moisture, since the opening time of stomata can be reduced. Therefore, the conditions for soil CH4 emissions are improved (Kim, 2013). However, the study by Wang et al. (2019) showed that the degradation of artificial clover meadow at a semi-arid loess hill site had a significant effect on the distribution and diversity of other herbaceous plants. This is the reason why the soil CH4 uptake of CK in this study was not significantly different from that of other plant community types (P > 0.05, Fig. 5). Differences in plant community type may lead to differences in soil compaction (i.e., STP), which affects the movement of water and gases, and thus the gas diffusion conditions for CH4 (Christiansen and Gundersen, 2011), with similar results presented in the current study (Table 2, Fig. 5).
In addition to proximal factors, there are a number of distal drivers that also influence soil CH4 fluxes (Robertson, 1989). The distinct differences in morphology of different vegetation conditions affect the variation in soil temperature by influencing light, wind speed, rainfall, etc. Among them, arboreal vegetation (XS) has the greatest advantage in microclimate regulation. The main source of energy for soil temperature is radiant energy from the sun, and tall arboreal vegetation (XS) shades the soil because of its higher leaf area and associated height (Kim, 2013; Oertel et al., 2016), which made the T10 significantly lower in XS than in HR and SB (Table 2; Figs. 2b and c). The water content of the soil in the study area is very low, and there is almost no free water; plus, the heat exchange between the soil and the environment is mediated by the air, and the soil temperature is closely linked to the movement of soil water. Therefore, the SWC10 of XS is significantly higher than that of other plant community types (Table 2; Figs. 2b and c). Vegetation caused differences in soil temperature and moisture, which in turn influenced the changes in soil CH4 flux. It has been shown that lower soil pH, which reduces the activity of soil microorganisms, leads to lower soil CH4 uptake (Gatica et al., 2020). However, in this study, lower variation in soil pH among the plant community types did not significantly affect methanotrophic bacteria or archaea, which is consistent with the findings of Werner et al. (2007) and Ai et al. (2021). On the other hand, Nitrogen (nitrate, ammonia) may have a positive or neutral effect on soil CH4 uptake in boreal temperate forest communities (Xia et al., 2020), which is similar to the results previously reported by our team in this region (Yan et al., 2019), albeit there is also no statistically significant relationship. It is well known that slope and slope orientation play a highly important role in regulating local temperature, radiation, and soil moisture. Among the five plant community types, XS, HR, and MX were located on shady slopes, CK on positive slopes, and SB in a no-slope orientation zone. The results showed that the soil CH4 fluxes were not significantly correlated with slope orientation, which is consistent with the findings of Fang et al. (2009). This may have been caused by the fact that all sampling plots were laid out on almost flat cultivated land after land preparation and that the meteorological conditions were essentially the same.
-
Changes in vegetation and its microenvironment also make soil CH4 fluxes show different temporal patterns of variation. In this study, the maximum values of soil CH4 uptake under different plant community types in LHR occurred in the growing season, and the minimum values all occurred in winter. This is due to the freezing of the soil in winter, which decreases soil pore connectivity (Bhandral et al., 2007) and reduces microbial activity, resulting in lower uptake rates (Do Carmo et al., 2012). On the one hand, as solar radiation increases, the soil thaws and the soil temperature increases, allowing an increase in the rate of gas diffusion and the probability of microorganisms obtaining contact with oxygen (Singh et al., 2010), which accelerates the rate of CH4 oxidation and increases the uptake of CH4 by the soil (Nesbit and Breitenbeck, 1992; Castro et al., 1995; Dou et al., 2015). Additionally, increased rainfall increases the soil water content, the CH4 oxidizing bacteria are transformed from a dormant to active state, and increasing soil water content within a certain range is beneficial for CH4 oxidative uptake (Chen et al., 2002). Due to the soil properties (Wu et al., 2018) and the climatic characteristics of low rainfall and high evaporation in the study area, higher transpiration after rainfall accelerates the oxidation and transportation of CH4 (Chen, 2003). However, it has also been shown that soil CH4 fluxes are not simply linearly related to soil moisture (Ju et al., 2016; Zhang et al., 2019), and when soil water content continues to increase, it will cause an increase in soil water-filled pore space, which limits the rate and diffusion of atmospheric CH4 oxidation (Werner et al., 2006; Curry, 2007; Wu et al., 2010). However, due to low rainfall and high evaporation in the study area, the SWC10 of the different plant community types could not be consistently increased to the saturated soil water content. Therefore, this idea could not be verified. Temperature and soil moisture are considered to be the main factors affecting the seasonal variation in soil CH4 fluxes (Song et al., 2009; Zhu et al., 2015a, b), and the effects of both interact with each other and show different dominance depending on the characteristics of the ecosystem (Olefeldt et al., 2013). This reason is also evidenced by the linear regression relationship between T10, SWC10, and soil CH4 fluxes in different plant community types in this study (Fig. 7), which is similar to the results of previous studies (Moore and Dalva, 1993; Glatzel et al., 2004; Cui et al., 2017; Ma et al., 2020). Furthermore, the fact that precipitation, as the main source of soil moisture in this area, is distributed in a pattern that is consistent with that of the temporal variation in soil CH4 flux, also reiterates this view (Figs. 2 and 6). The results are similar to the findings of other studies (Ju et al., 2016; Ma et al., 2020). In contrast, the maximum uptake value at the MS site occurred in early spring; due to the enclosed setup of the MS site, the aboveground plants that had died out in the previous year’s non-growing season increased the soil temperature (Li et al., 2018), thus increasing the uptake of CH4 by the soil. With the growth of surface vegetation, the ground vegetation cover gradually increased, due to which the soil temperature gradually decreased, thus causing a change in soil CH4 uptake in the form of an increase first, and then a decrease. Regarding the cumulative flux of soil CH4, Wang et al. (2014) showed that the average rate of soil CH4 uptake in Chinese forest and grassland was 49.43 μg m−2 h−1 and 59.62 μg m−2 h−1, respectively, with annual cumulative fluxes of 4.22 kg ha−1 and 5.09 kg ha−1. These data are comparable to the rate of soil CH4 uptake in other forest and grassland sites worldwide (Wang et al., 2014). The average annual cumulative soil CH4 uptake fluxes of the different plant community types in this study were 2.02 and 1.67 times higher than the annual cumulative uptake fluxes of Chinese forest and grassland, respectively, when only vegetation was considered. Therefore, it can be seen that the soil CH4 uptake of different plant community types in this region is much higher than the average value in China, which is important for CH4 reduction and mitigation of the greenhouse effect.
-
The results of the study showed that all five plant community types were sinks of soil CH4 in LHR. Different plant community types significantly affected soil CH4 fluxes in decreasing order as follows: SB > HR > CK > MS > XS. Throughout the study period, the maximum values of soil CH4 uptake occurred in the growing season, while the minimum values all occurred in winter. Soil CH4 fluxes were significantly and positively correlated with soil temperature and soil moisture. The combined effect of soil temperature and moisture is an important controlling factor for the temporal variability of soil CH4 fluxes. In LHR, Stipa bungeana grassland is the plant community type with the highest capacity for soil CH4 uptake among the five plant community types studied, and is a more desirable plant community type for mitigating soil CH4 emissions. In the process of vegetation restoration in LHR, the soil CH4 absorption potential of different plant community types should be considered, ecological benefits should be taken into account, and vegetation more suitable for mitigating the greenhouse effect should be selected. Therefore, when estimating the soil CH4 exchange in LHR, it is necessary to consider the influence of the different plant community types.
Acknowledgements. We would like to thank all the people who were involved in the field and laboratory work. This study was financially supported by the Gansu Province Key Research and Development Program (Grant No. 20YF8NA135), the Gansu Province Financial Special Project (Grant No. GSCZZ 20160909), and the Industrial Support Program Project (Grant No. 2021CYZC-15, No.2022CYZC-41). The authors would like to thank Director Jin CHEN, Xiaoping WANG, and other staff from the Soil and Water Conservation Research Institute in Dingxi City, Gansu Province, for their support with the management of the experimental field, and Bin WU of the Ecological Research Center of Northeast Forestry University for his advice on the experimental data in this paper.
| Plot | Area (m2) | Longitude and latitude | Elevation (m) | Main species | Coverage (%) | Height (cm) |
| MS | 20 × 20 | 104°39′1.82′′E 35°34′48.07′′N | 1990 | Medicago sativa (90%), Stipa bungeana, Artemisia lavandulifolia | 90.06±1.58b | 62.23±0.23d |
| XS | 20 × 20 | 104°39′10.62′′E 35°34′45.08′′N | 2018 | Xanthoceras sorbifolium (86%), Bupleurum chinense, Gentiana macrophylla, Leontopodium leontopodioides | 95.32±2.35c | 76.45±2.54e |
| CK | 20 × 20 | 104°39′1.51′′E 35°34′45.00′′N | 1999 | Caragana korshinskii (42%), Potentilla chinensis, Picris hieracioides | 50.43±5.45a | 16.00±0.79a |
| HR | 20 × 20 | 104°39′0.18′′E 35°34′47.32′′N | 1998 | Hippophae rhamnoides (81%), Leontopodium leontopodioides, Artemisia annua | 89.26±0.78b | 54.89±1.23c |
| SB | 20 × 20 | 104°39′3.05′′E 35°34′45.48′′N | 2008 | Stipa bungeana (92%), Plantago asiatica, Setaria viridis, Leymus secalinus | 98.23±0.63c | 35.93±0.44b |
| Notes: XS, Xanthoceras sorbifolium forestland; SB, Stipa bungeana grassland; CK, Caragana korshinskii bushland; HR, Hippophae rhamnoides shrubland; MS, Medicago sativa grassland. The percentage of the main species is the coverage of the dominant species. Values are presented as mean ± standard error. Superscript letters indicate significant differences between plant community types (P < 0.05). | ||||||




 DownLoad:
DownLoad:












 AAS Website
AAS Website 
 AAS WeChat
AAS WeChat